Recently, several tissue clearing methodologies have been developed that render organs transparent and allow section-free imaging of significant volumes, thereby improving our capacity to study the relationships between cell positioning and 3D tissue architecture ( 1– 11). Finally, many cell types require simultaneous visualization of multiple phenotypic markers for correct subset identification, making interpretation of cell composition within tissues problematic without the use of highly multiplexed imaging panels. 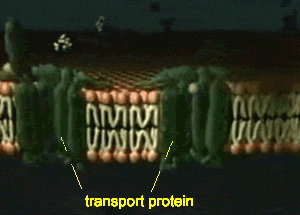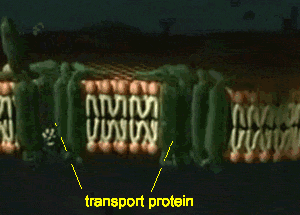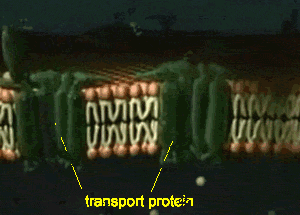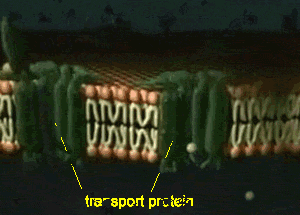
Furthermore, detection and analysis of rare cellular events requires imaging of a large number of disconnected sections, which introduces possible image selection bias and suffers from the potential omission of key physiological landmarks located just outside of the sampled area. Although providing an excellent framework for understanding general features and the respective positioning of well-represented cell types, such data lack information on 3D organization, being particularly limiting for irregular structures such as the vasculature, airways, nervous tissue, inflamed sites, tumors, or reactive lymph nodes. The majority of information on such processes comes from microscopic imaging of relatively thin (5–20 μm) “2D” tissue cross-sections, examining several markers of interest to visualize a limited number of cell populations with respect to a tissue’s representative structural elements. Combined with histo-cytometry, C e3D provides a comprehensive strategy for volumetric quantitative imaging and analysis that bridges the gap between conventional section imaging and disassociation-based techniques. We use this technology to demonstrate large-volume, high-resolution microscopy of diverse cell types in lymphoid and nonlymphoid organs, as well as to perform quantitative analysis of the composition and tissue distribution of multiple cell populations in lymphoid tissues. This enhanced signal quality and capacity for extensive probe multiplexing permits quantitative analysis of distinct, highly intermixed cell populations in intact C e3D-treated tissues via 3D histo-cytometry. Here we report an easy to use method, clearing-enhanced 3D (C e3D), which generates excellent tissue transparency for most organs, preserves cellular morphology and protein fluorescence, and is robustly compatible with antibody-based immunolabeling. For this reason, comprehensive, high-resolution mapping of cell positioning, phenotypic identity, and functional state in the context of macroscale tissue structure is critical to a deeper understanding of diverse biological processes. Resting state is when membrane potential returns to the resting voltage that occurred before the stimulus occurred.įor more information, please register to download our Guide, the Axon Guide.Organ homeostasis, cellular differentiation, signal relay, and in situ function all depend on the spatial organization of cells in complex tissues.Hyperpolarization is a lowered membrane potential caused by the efflux of potassium ions and closing of the potassium channels.Membrane Repolarization results from rapid sodium channel inactivation as well as a large efflux of potassium ions resulting from activated potassium channels.Depolarization is caused by a rapid rise in membrane potential opening of sodium channels in the cellular membrane, resulting in a large influx of sodium ions.In patch-clamp mode, sufficient current must be administered to the cell in order to raise the voltage above the threshold voltage to start membrane depolarization.  
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
